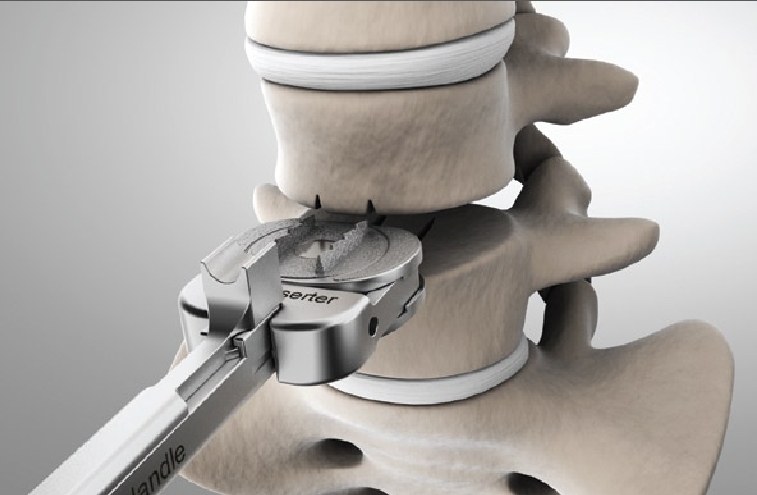
Spinal stenosis is the compression of nerve roots by bone or soft tissue secondary to the narrowing of the spinal canal, lateral recesses, or intervertebral foramina. Spinal stenosis may have acquired or congenital origins. Most cases are acquired and caused by hypertrophy of the ligamentum flavum, enlarged osteophytes, degenerative arthritis, disk herniations, and various systemic illnesses. The ligamentum flavum (LF) is a highly specialized elastic ligament that connects the laminae of the spine and fuses them to the facet joint capsules. There are a number of treatment options available for spinal stenosis. Implants and surgical interventions have grown in popularity recently, and a number of these have been shown to have varying efficacy, including the minimally invasive lumbar decompression (MILD®), Vertiflex®, Coflex® Interlaminar Stabilization, and MinuteMan G3® procedures. Minimally invasive lumbar decompression (MILD®) is a minimally invasive outpatient procedure to treat spinal stenosis related to hypertrophied ligamentum flavum. The Superion® Interspinous Spacer, also known as Vertiflex®, is a titanium implant that is delivered percutaneously to relieve back pain caused by lumbar spinal stenosis. The MinuteMan® is a minimally invasive, interspinous-interlaminar fusion device planned for the temporary fixation of the thoracic, lumbar, and sacral spine, which eventually results in bony fusion. Based on our review of the available current scientific literature, the novel interventions for symptomatic lumbar spinal stenosis, such as the MILD® procedure and the Superion® interspinous spacer, generally appear to be safe and effective. There is a possibility in the future that these interventions could disrupt current treatment algorithms for lumbar spinal stenosis.
Keywords:
Claudication; Coflex® interlaminar stabilization; Hypertrophy ligamentum flavum; MILD® procedure; MinuteMan G3®; Spinal stenosis; Superion® interspinous spacer; Vertiflex®.